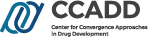RESEARCH
- Microtracing/microdosing Study
- Use of Artificial Intelligence for Clinical Drug Development
- Policy Analysis for an Integrated Clinical Research System in Korea
- Systems Pharmacology and Pharmacometrics
- Writing an Introductory Book on Biologics
- Current status of integration of EMR data in Korea
- Technologies available for integration of public health data and hospital EMRs
- Availability of a metadata model for integrated clinical research systems in other countries
- Roles and responsibilities of the centralized coordinating center for integrated clinical research systems
Policy Analysis for an Integrated Clinical Research System in Korea
CCADD is studying the strategic policy issues to efficiently and seamlessly integrate standalone clinical research systems in Korea such as drug- or disease-registries (public or private), open databases created by the government, and hospital’s electronic medical records (EMRs). This study is sponsored by the Korea National Enterprise for Clinical Trials (KoNECT).
In this study, CCADD has first enumerated the available clinical trial resources in Korea followed by a systematic investigation of the current legal, societal, and cultural frameworks that have prevented clinical trial systems being fully integrated. CCADD has also studied the following topics:
The results of this study will be published as a white paper around the end of 2017.



CCADD
Center for Convergence Approaches in Drug Development, Graduate School of Convergence Science and Technology, Seoul National University
Room C-208, 145 Gwanggyo-ro, Yeongtong-gu, Suwon-si, Gyeonggi-do, 16229, SOUTH KOREA (Gwanggyo)
Room 406, Building 17, Seoul National University College of Medicine, 103 Daehak-ro, Jongno-gu, Seoul, SOUTH KOREA (Yeon-gun)
Tel: +82-31-888-9189 (Gwanggyo); +82-2-3668-7381 (Yeon-gun)
Fax: +82-31-888-9575
Email: ccadd.snu@gmail.com