RESEARCH
- Microtracing/microdosing Study
- Use of Artificial Intelligence for Clinical Drug Development
- Policy Analysis for an Integrated Clinical Research System in Korea
- Systems Pharmacology and Pharmacometrics
- Writing an Introductory Book on Biologics
Writing an Introductory Book on Biologics
Over the last decade, we have witnessed how technology innovations, such as smartphones and artificial intelligence, can quickly change our lives. The biopharmaceutical industry is not an exception. Since the human genome project was successfully completed, the cost of genetic analysis has rapidly decreased, making genetic analysis more affordable for routine patient care. Furthermore, personalized medicine combined with artificial intelligence technology shed bright light on the possibility of reanalyzing the relationship between patients and diseases.
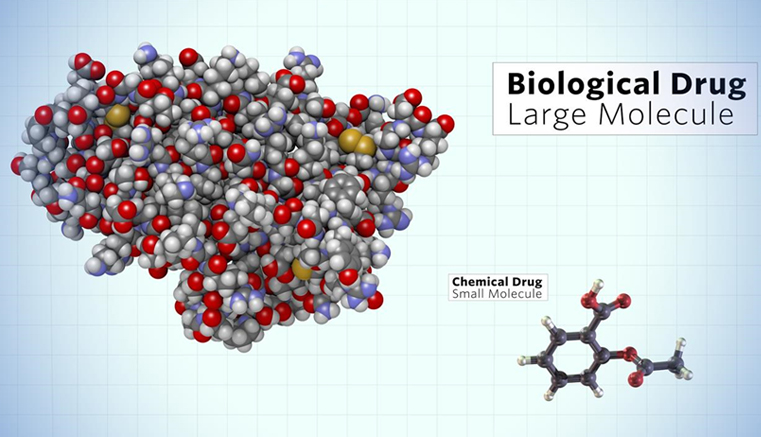
More extensive development and use of biological agents or biologics based on advanced medical technologies is one of those new trends. To support this notion, it is estimated that almost 50% of research expense by big pharmaceutical companies has been spent in the development of biologics. Biologics currently occupy approximately a quarter of the total pharmaceutical market in terms sales, which is on the sharp rise. For example, seven out of the top 10 best-selling drugs in 2016 were biologics.
However, lay people know little about biologics. This is rather odd because Humulin, the first biopharmaceutical developed by Genentech, was approved by FDA more than 35 years ago (1982). Even OKT3, the first monoclonal antibody drug, was first approved in 1986. Their lack of appreciation on biologics may have something do with the fact that the development and production of biologics involves a variety of modern biological disciplines including molecular biology, to which lay people are not easily accessible.
Based on this understanding, we decided to write an introductory book on biologics that may help lay people understand the core principles of biologics and their follow-on drugs or biosimilars in association with their development, manufacturing, and regulatory implications. We hope that the readers will be able to understand the dynamic changes that are happening in the course of developing biologics and the marketplace. The book is expected to be published late 2017.



CCADD
Center for Convergence Approaches in Drug Development, Graduate School of Convergence Science and Technology, Seoul National University
Room C-208, 145 Gwanggyo-ro, Yeongtong-gu, Suwon-si, Gyeonggi-do, 16229, SOUTH KOREA (Gwanggyo)
Room 406, Building 17, Seoul National University College of Medicine, 103 Daehak-ro, Jongno-gu, Seoul, SOUTH KOREA (Yeon-gun)
Tel: +82-31-888-9189 (Gwanggyo); +82-2-3668-7381 (Yeon-gun)
Fax: +82-31-888-9575
Email: ccadd.snu@gmail.com